Fluid and electrolyte review on hypochloremia and hyperchloremia for nursing students!
This review is part of a comprehensive fluid and electrolyte series. In this review you will learn the causes, signs/symptoms, and nursing interventions associated with hypo and hyperchloremia.
Don’t to access the free hypochloremia and hyperchloremia quiz when you’re done reviewing this material.
Nurse Sarah’s Notes and Merch

Just released is “Fluid and Electrolytes Notes, Mnemonics, and Quizzes by Nurse Sarah“. These notes contain 84 pages of Nurse Sarah’s illustrated, fun notes with mnemonics, worksheets, and 130 test questions with rationales.
You can get an eBook version here or a physical copy of the book here.
Chloride
What is Chloride?
Chloride is an electrolyte that has an important relationship with other electrolytes, especially sodium. Chloride is a negatively charged ion, while sodium is positively charged, and they both like to congregated outside the cell. Therefore, because of this, usually if there is a loss of sodium, there will be a loss of chloride (vice versa). In turn, there is some overlapping with the causes, signs/symptoms, and interventions.
Chloride also has an opposite relationship with bicarb (when chloride is low, bicarb is high etc.) in part because how there is an exchange in the red blood cell for proper gas exchange.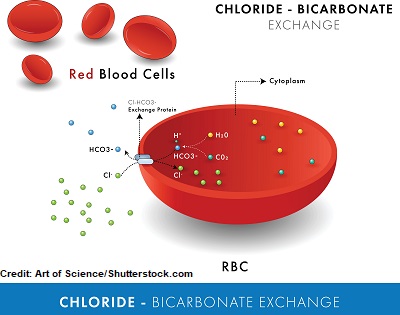
Furthermore, when an imbalance of chloride occurs due to either metabolic acidosis or alkalosis, the potassium level will be altered as well. So, that is why as the nurse when you’re seeing a chloride imbalance you may also want to look at the sodium, bicarb and potassium levels.
Chloride plays a role with acid-base balance in the body along with balancing the fluids in our body by working with sodium to maintain osmotic pressure. Chloride is also needed to make hydrochloric acid, which plays a huge role in food digestion. The kidneys help maintain chloride blood levels by taking what you don’t need and excreting it in the urine. Chloride is also excreted through sweating and gut juices.
Therefore, if chloride is imbalanced (especially high) it may be a kidney problem because specific parts of the nephron remove chloride from the blood and place it in the urine (if the kidneys aren’t functioning right this can’t happen so more stays in the blood).
Normal level: 95-105 mEq/L
Hypochloremia
Low chloride levels in the blood ( <95 mEq/L)
Causes:
- Loss of Chloride: GI system (vomiting, gastric juice loss via NG tube suction or ileostomy)
- Diuretics (thiazides and loop diuretics affect how renal tubules reabsorb sodium and chloride which causes more loss of the ions in the urine)
- Burns
- Cystic fibrosis (high amounts of chloride in the sweat)
- Metabolic alkalosis (increase in bicarbonate leads to low levels of chloride…they have an opposite relationship due to how each ion shifts in the red blood cells for proper gas exchange)
- Fluid overload (diluting extracellular fluid: CHF, SIADH (Syndrome of Inappropriate Antidiuretic Hormone Secretion): too much ADH is released causing the body to retain water, which dilutes the sodium level)
Signs and symptoms of hypochloremia tend to be associated with whatever is causing the low level (rather than the level being low itself, so you want to look at what is causing the low level to identify the signs and symptoms) and tends to look like hyponatremia signs and symptoms but you may see:
- Dehydration (increased heart rate, decrease blood pressure, fever)
- Vomiting, diarrhea, lethargic
Nurse’s Role & Treatment for Hypochloremia
LOSS
Look at the patient’s sodium level and assess for signs/symptoms of hyponatremia (normal 135-145 mEq/L).
Remember “SALT LOSS” for signs associated with hyponatremia
- Seizures & Stupor (decrease in consciousness…confusion)
- Abdominal cramping
- Lethargic
- Tendon reflexes diminished, trouble concentrating
- Loss of urine & appetite
- Orthostatic hypotension, overactive bowel sounds
- Shallow respirations (happens late due to skeletal muscle weakness)
- Spasms of muscles
In addition, monitor neuro status (patients can become very confused and are at risk for injury…this confusion can be from swelling in brain), seizure precautions, respiratory status, I and O’s, VS (especially blood pressure), weights
Other labs to monitor:
- high bicarb and low potassium (especially if this is due to metabolic alkalosis)
- bicarb and potassium are also related to the balance of chloride because they all work together to balance the acid-base system and our fluid balance in the body
Saline (normal saline 0.9%) IV administration to replace chloride
Sources of chloride rich foods
- table salt, tomatoes, olives, seafood, processed meats and canned foods
Hyperchloremia
High chloride in the blood ( >105 mEq/L)
Causes:
- High intake of sodium (IV fluids…hypertonic fluids or too much saline)
- Not drinking enough water or losing too much water (vomiting, diabetes insipidus: urinating so much fluid, sweating)
- Losing too much bicarb via diarrhea
- Metabolic acidosis (kidney failure or medications that cause it)
- Hyperaldosteronism (Conn’s Syndrome)…too much production of aldosterone (retain sodium along with fluid and excrete potassium)
- Corticosteroids
Signs and Symptoms of hyperchloremia tend to be associated with whatever is causing the high level (rather than the level being high itself) so you want to look at what is causing the high level to identify the signs and symptoms) and it may look like hypernatremia and acidosis signs and symptoms.
Signs/Symptoms of Hypernatremia that could present with hyperchloremia….remember FRIED
- Fatigue
- Restless, really agitated (confused….central nervous system changes)
- Increased reflexes, respirations (progress to seizures and coma)
- Extreme thirst (*big sign)
- Decreased urine output, dry mouth/skin
Nurse’s Role and Treatment for Hyperchloremia
HI CL-
Hold sodium chloride infusions (further increase levels of chloride) along with sodium and chloride rich foods (needs low sodium diet)
Instead Lactated Ringers may be used to help decrease chloride
Why? The lactate is turned into bicarb, which helps increase bicarb levels and lower chloride (remember bicarb and chloride have an opposite relationship)
- If the cause of hyperchloremia is metabolic acidosis the increase in bicarb will help increase the pH in the blood
- In addition, the administration of bicarb and certain diuretics can help decrease the chloride level too.
Collect I and O’s, weights, vs
Labs to monitor:
- Chloride
- Sodium
- Bicarb (make sure not becoming too alkaline and blood pH increasing too much)
- Potassium (hyperkalemia…if acidosis presents potassium leaves the cell and moves into the extracellular (hence blood) in exchange for hydrogen ions)
References:
Chloride in diet: MedlinePlus Medical Encyclopedia. Medlineplus.gov. (2021). Retrieved 29 November 2021, from https://medlineplus.gov/ency/article/002417.htm.
Shrimanker I, Bhattarai S. Electrolytes. [Updated 2021 Jul 26]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK541123/