Understanding osmosis and osmolarity can help you understand how IV fluids can be useful in healthcare. Let’s take a look at some of the key concepts of osmosis and osmolarity.
Osmosis and Osmolarity
- A passive type of water transport
- Water molecules move through a semi-permeable membrane that only allows water through.
Look at osmosis these two ways:
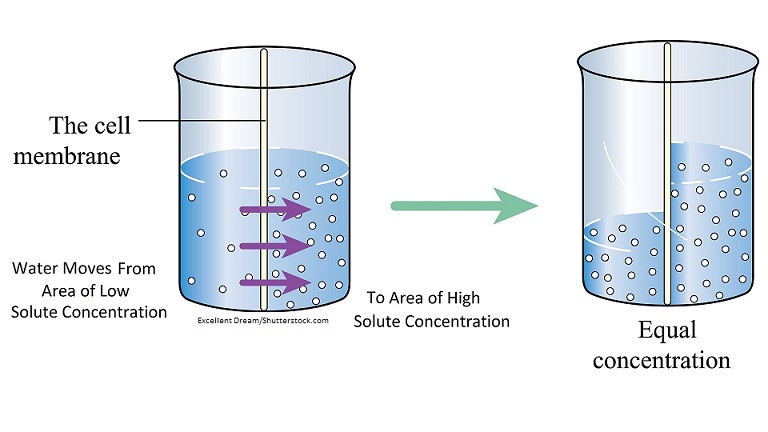
Water will move from a fluid of a higher water concentration to a fluid of lower water concentration.
OR
Water will move from a lower solute concentration fluid to a higher solute concentration fluid.
Simplified: Water is drawn to the fluid that has the MOST solutes.
What is a solute?
A solute is a solid dissolved in a liquid.
Many substances can be a solute. For example, sodium and chloride are solutes in a bag of IV fluids. They are also considered electrolytes in this form.
The liquid or the solution that contains the solutes is known as the solvent.
Osmosis is influenced by how concentrated a fluid is of solutes, hence its osmolarity.
Osmolarity
Osmolarity is the total concentration of solutes in a solution (per liter).
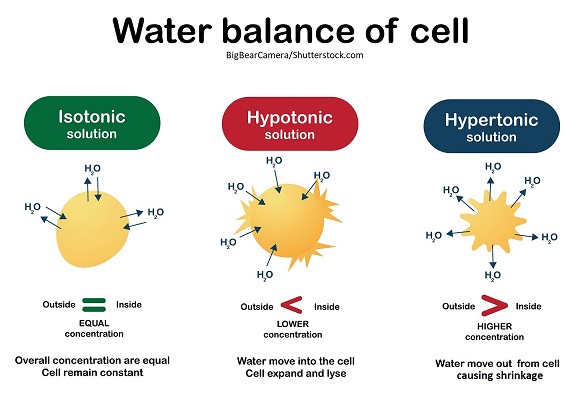
IV fluids can have an equal, high, or low osmolarity when compared to the blood plasma.
High osmolarity = high amount of solutes in a solution but low amount of water
Example: hypertonic solutions
Low osmolarity = low amount of solutes in a solution but high amount of water
Example: hypotonic solutions
Connecting the dots:
We can use the principles of osmosis and osmolarity to help treat patients who need fluids replaced based on what fluid compartment needs to be treated by shifting fluids around. This can be done by administering various types of IV fluids that have different osmolarities or solute concentrations, which will move water in or out of these fluid compartments.